Stabilizing the adaptive internal compass of a migrating neutrophil
01 — Objective: To demonstrate how thorough examination and economy of language can intersect to form a precise account of a complex cellular mechanism within strict word limits.
02 — Material:
Molecular working memory: septins stabilize the adaptive internal compass of a migrating neutrophil
SPECIFIC AIMS During chemotaxis, neutrophils must be responsive to target signals as to converge on sites of infection, without being diverted by excessive sensitivity to the barrage of sensory noise in the animal body. Equal to the task, motile immune cells perform gradient tracking with lethal accuracy, operating just above the theorized limit of detection. This vital balance in responsiveness is calibrated in part by a transient memory of directionality that is sustained by the cytoskeletal architecture even as intracellular signaling fluctuates alongside dynamic sensory input.
In a 3D environment, neutrophil polarity is intimated by the extension of a microtubule-scaffolded, actin-rich protrusion, called a pseudopod, up the chemotactic gradient. Pseudopodia potentiate forward migration by extending and retracting in a coordinated fashion at the leading edge of the neutrophil. As this unfolds, each nascent pseudopod extends in the same position as the one antepenultimate, indicating a molecular memory of recent position that is stored and acted upon by the cell. Existing evidence suggests that a structural cue for this memory is the local curvature of the membrane at the site of extension. Although 3D chemotactic migration is a quintessential course of movement for neutrophils in vivo, it is not fully understood. The structures involved in capturing provisional record of protrusion sites remain poorly characterized, and it is yet unknown how neutrophils then act on this temporarily stored information to target re-extension at the specific, indicated position.
Septins
are a family of GDP-binding proteins that can form a variety of
higher order structures and have the intrinsic ability to
differentially bind to distinct radii of membrane curvature with micrometer-scale precision. Furthermore, septins are known to indirectly and directly interact
with actin filaments and microtubules as key mediators in an array of processes. We
hypothesize that septins help stabilize an acting cellular memory of pseudopod
position by binding to curvature sites induced in the membrane during
extension, and that membrane-bound septins subsequently help
coordinate re-extension by capturing and precipitating site-specific plus
end growth of microtubules.
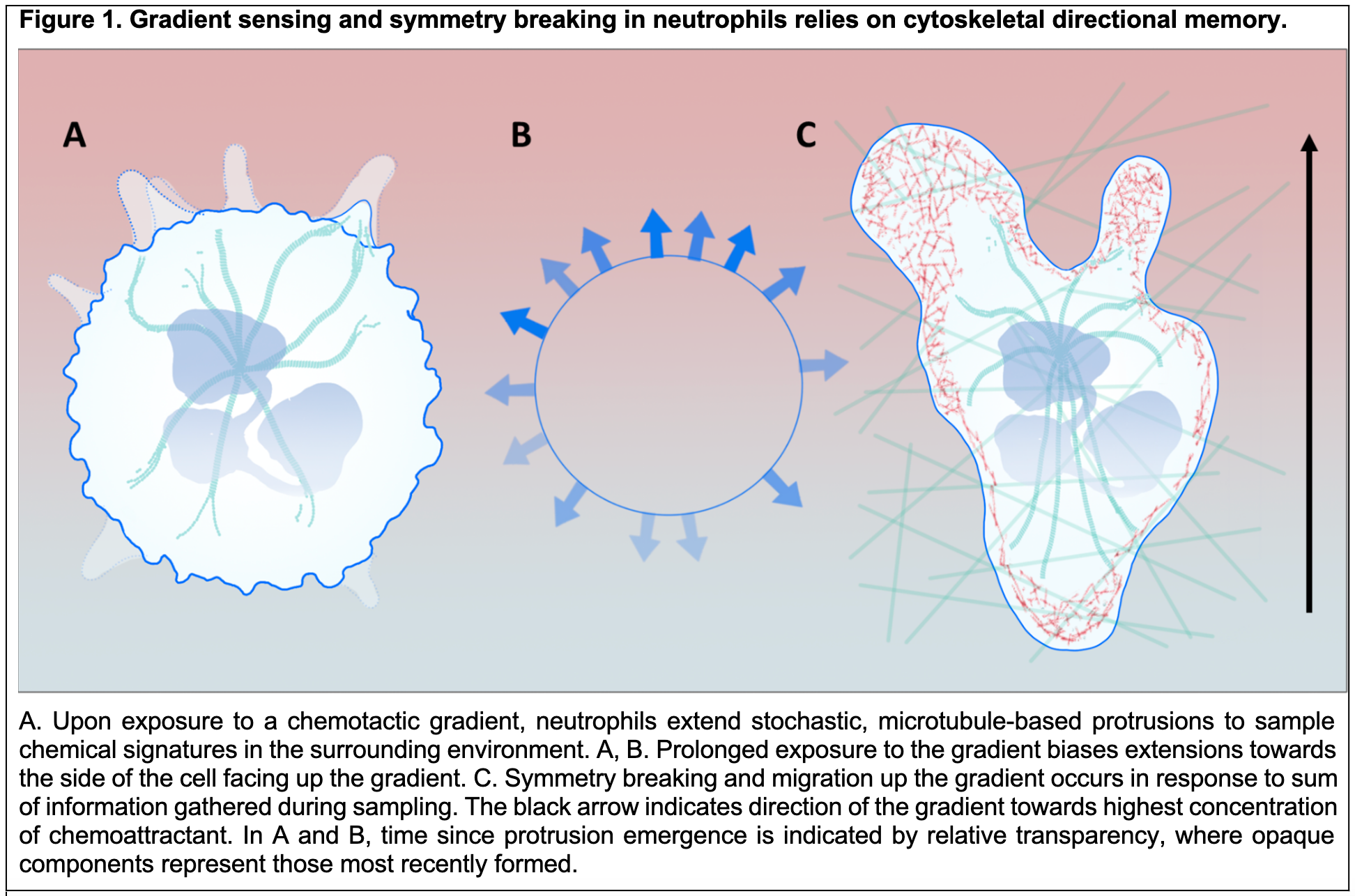
BACKGROUND Storage and transmission of information is a fundamental process of living systems. In addition to long-term keeping of information, such as that encoded in the genome, cells are constantly integrating and responding to dynamic sensory input, necessitating short-term information storage -- or metaphorical "molecular working memory.” Neutrophils, part of the cellular innate immune response against pathogens, are remarkable in their ability to accurately navigate a changing information landscape. These cells rapidly converge on the site of a wound or infection through chemotaxis, or movement in response to an extracellular chemical gradient. This process is fine-tuned at each step so that the system remains adaptive, but not so reactive that signal tracking is derailed by fluctuations in the body that may momentarily eclipse the primary signal. Such precise regulation levies the diverse inherent tendencies of multiple systems within the cell. Thus, while signaling cascades may waver, the cytoskeletal architecture provides a more stable record of direction and polarity.
During initial gradient sensing, dynamic instability of microtubules drives the stochastic emergence and retraction of protrusions from the membrane. These protrusions or microtentacles sample the chemical environment on all sides of the cell. In the presence of a persistent chemical gradient, frequency of protrusions is biased toward the side that is enduring the greatest concentration of chemoattractant. During polarity establishment and symmetry breaking in a 3D environment, the cumulative cellular memory of protrusion sites culminates in the formation of a pseudopod that faces up the gradient (Figure 1). As migration begins, extension of pseudopodia is coordinated such that every other extension will occur at the same site of the membrane as the one before the previous. Experimental evidence has indicated that cells record a memory of position (as opposed to a memory of direction), and that the sensory cue for this information is most likely local membrane curvature. However, it is unknown how cells maintain a molecular memory of the site of protrusions and act on this memory to drive re-extension at the same site.
Septins, a family of GTPases sometimes referred to as the fourth component of the cytoskeleton, are compelling candidates for the driver of this molecular memory of position. Higher order septin structures can stably persist over relatively long periods when compared to other cytoskeletal structures. They have the unique inherent ability to sense and assemble on membranes of specific positive curvatures. As such, the septin cytoskeleton is uniquely poised to record cellular memory of membrane position. Indeed, septins have been shown to perform similar functions in a variety of other cell types, notably, septins record a molecular memory of axon position that persists even after neuronal cell division.
We hypothesize that septins record a
cellular memory of positions during gradient sensing, and that this
is achieved through their innate ability to sense and stably bind to
distinct nanometer scale curvatures at the cell cortex. Septins also
likely mediate “recollection” of membrane position by capturing,
organizing, and mediating plus end growth of microtubules at
protrusion sites.

03 — Impact: Anchored a proposed research avenue within a broader conceptual landscape and positioned its central hypotheses to bridge the gaps in collective understanding of neutrophil movement.
Post a comment